43 identify the parts of the energy diagrams
Identify the four parts of the potential energy diagram. Identify the four parts of the potential energy diagram. See answers Advertisement Alleei Answer : The potential energy diagram is shown below. Explanation : Activation energy : The energy required to initiate the reaction is known as activation energy. Or, it is the amount of energy required to reach the transition state. 12.7 Catalysis | Chemistry - Lumen Learning Example 1: Using Reaction Diagrams to Compare Catalyzed Reactions. The two reaction diagrams below represent the same reaction: one without a catalyst and one with a catalyst. Identify which diagram suggests the presence of a catalyst, and determine the activation energy for the catalyzed reaction:
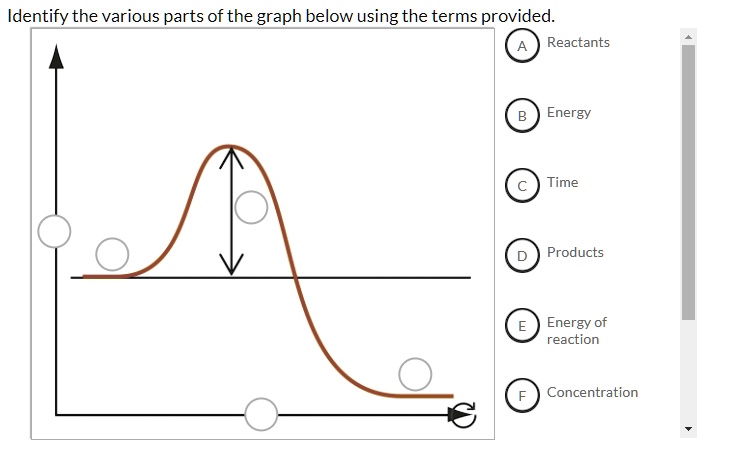
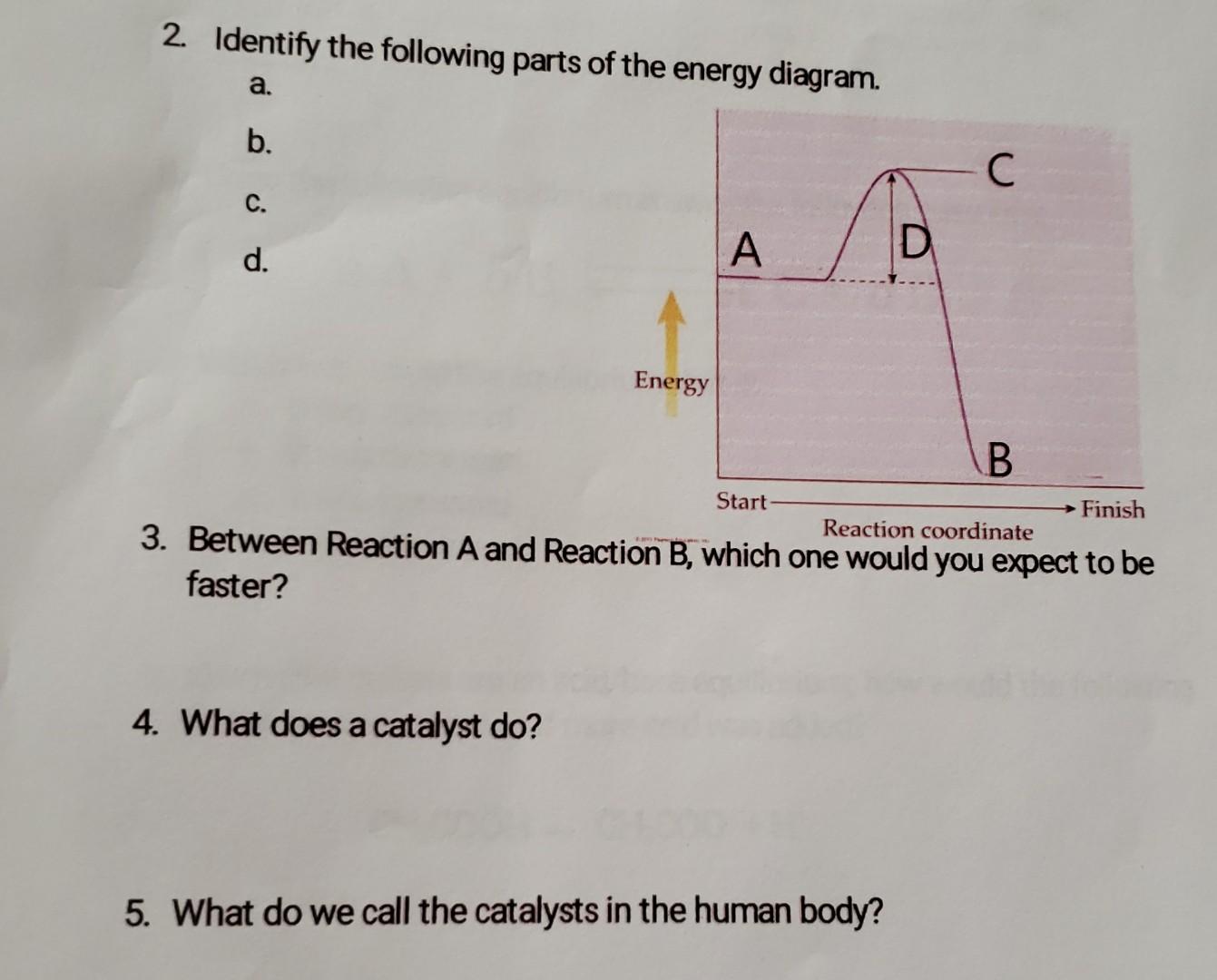
Identify the parts of the energy reaction coordinate | Chegg.com Answer to Identify the parts of the energy reaction coordinate
Identify the parts of the energy diagrams
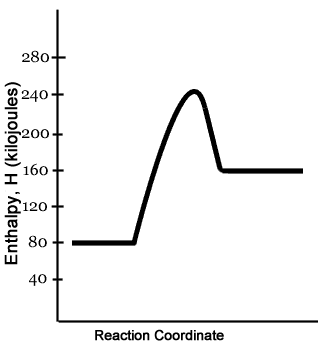
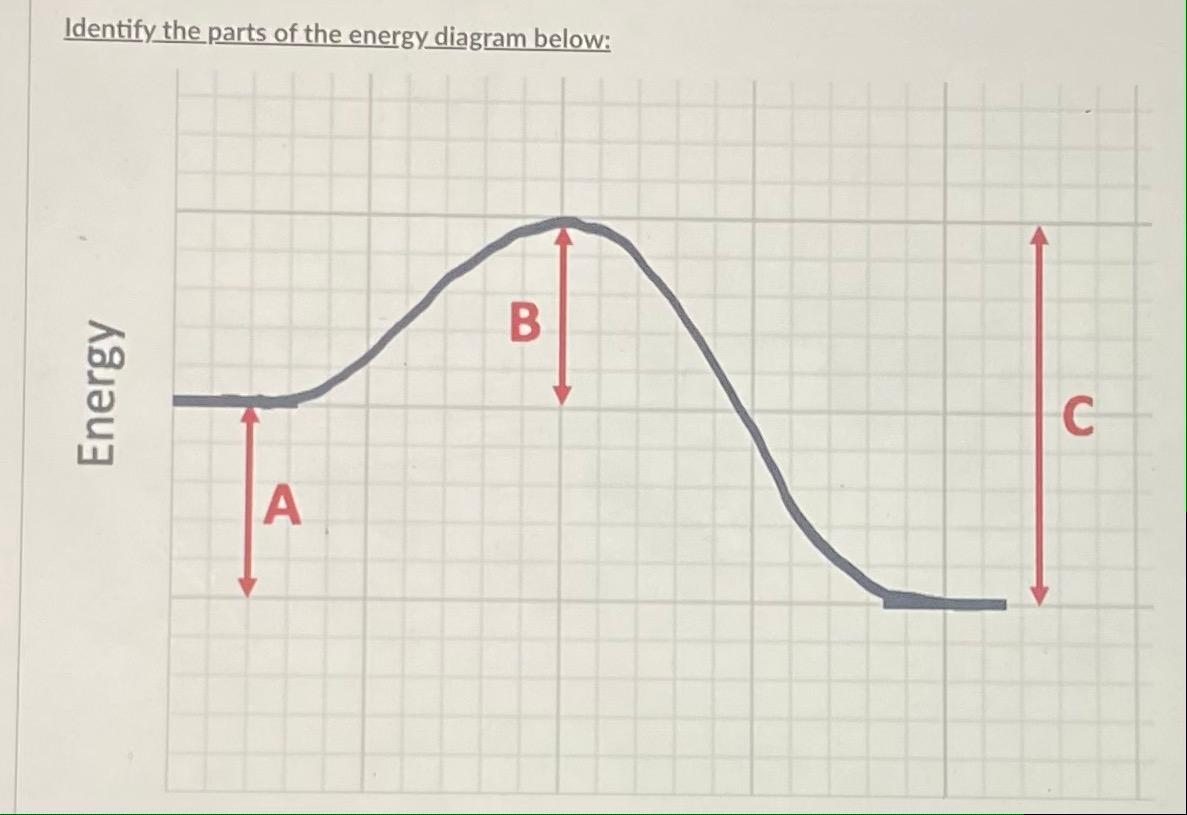
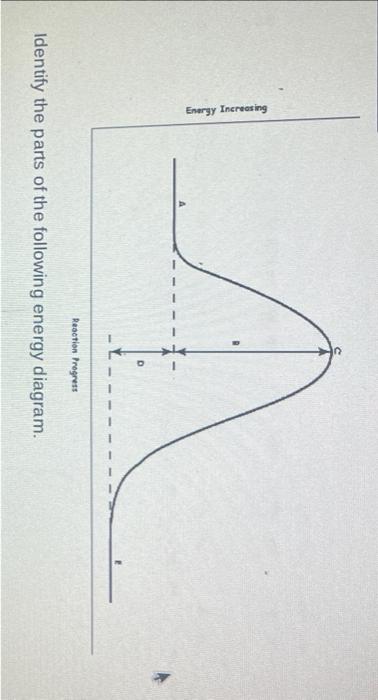
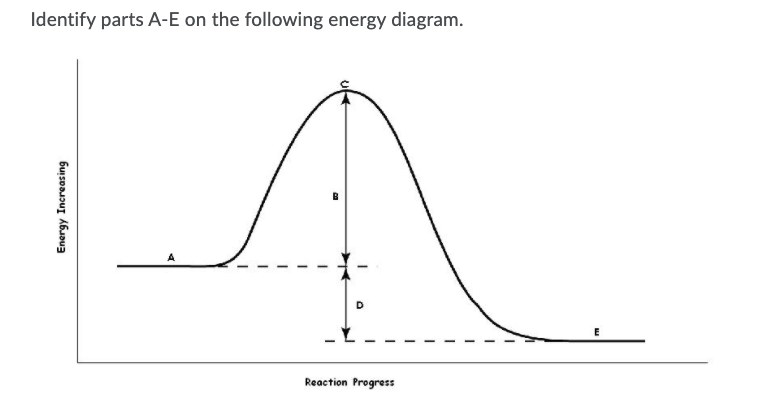
Energy Profiles (Energy Diagrams) Chemistry Tutorial - AUS-e-TUTE From our energy profile diagram we see that 192.4 kJ mol -1 of energy was absorbed by the reactant molecules, but only 100 kJ mol -1 was released as the activated complex broke apart to make the product molecules. Overall, the system absorbed a net amount of energy of 192.4 - 100 = 92.4 kJ mol -1 . Solved Identify the parts of the energy diagram below: B - Chegg Transcribed image text: Identify the parts of the energy diagram below: B Energy A C Reaction Progress Activation Energy (E₂): [Select] Heat of the Reaction (AHrxn): [Select ] Use the diagram to answer the questions: Is the value of AHrxn positive or negative? [Select ] Is this reaction endothermic or exothermic? Potential Energy Diagrams Flashcards | Quizlet is the difference between the potential energy of the reactants and products. The symbol for the heat of reaction is. Delta H. In a chemical reaction, an enzyme acts as a catalyst, causing the. activation energy of the reaction to increase. The only difference between a reaction with a catalyst and a reaction without a catalyst is.
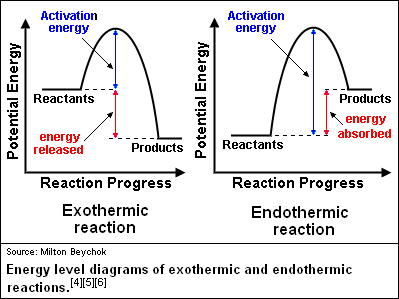
Identify the parts of the energy diagrams. Exothermic and endothermic reactions - AQA - BBC Bitesize The overall change in energy in a reaction is the difference between the energy of the reactants and products. Exothermic reactions The diagram shows a reaction profile for an exothermic reaction. 18.4: Potential Energy Diagrams - Chemistry LibreTexts The energy changes that occur during a chemical reaction can be shown in a diagram called a potential energy diagram, or sometimes called a reaction progress curve. A potential energy diagram shows the change in potential energy of a system as reactants are converted into products. identify the parts of the energy diagram - Brainly.com Answer : Graph 1 is an endothermic reaction and Graph 2 is an exothermic reaction. The labeling of both the graphs are shown below. Explanation : Transition state : When the reactant molecules absorb energy, their bonds are loosened and new lose bonds are formed between them. Solved Identify the energy associated with each of the - Chegg Question: Identify the energy associated with each of the labeled parts of the following diagram Progress of Reaction Drag the appropriate labels to their respective targets. Reset Help energy of products Region A Region B Region C Region D heat of reaction activation energy energy of reactants Show transcribed image text Expert Answer
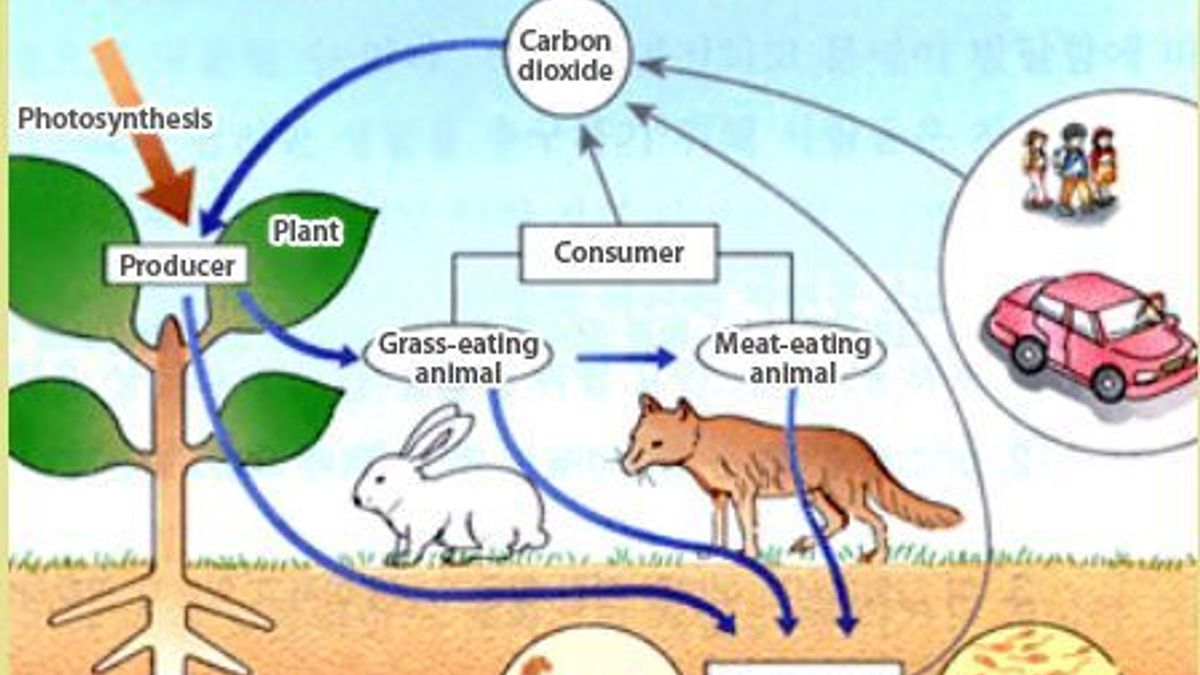
Reaction Mechanisms and Multistep Reactions - Chemistry LibreTexts Multi-step (Consecutive) Reactions. Mechanisms in which one elementary step is followed by another are very common. (step 1) A + B → Q. (step 2) B + Q → C. (net reaction) A + 2 B → C. (As must always be the case, the net reaction is just the sum of its elementary steps.) In this example, the species Q is an intermediate, usually an ... 5.6: Reaction Energy Diagrams and Transition States The activation energy is what determines the kinetics of a reaction: the higher the energy hill, the slower the reaction. At the very top of the energy barrier, the reaction is at its transition state (TS), which is the point at which the bonds are in the process of breaking and forming. The transition state is an ' activated complex': a ... Potential Energy Diagrams - Kentchemistry.com A potential energy diagram plots the change in potential energy that occurs during a chemical reaction. This first video takes you through all the basic parts of the PE diagram. Sometimes a teacher finds it necessary to ask questions about PE diagrams that involve actual Potential Energy values. Cell parts and functions (article) | Khan Academy Cells contain parts called organelles. Each organelle carries out a specific function in the cell. A cell's organelles work alone and together to keep the whole cell functioning. Mitochondria are organelles that break down sugars. This process releases energy that the cell can use. The nucleus is an organelle that contains a cell's genes.
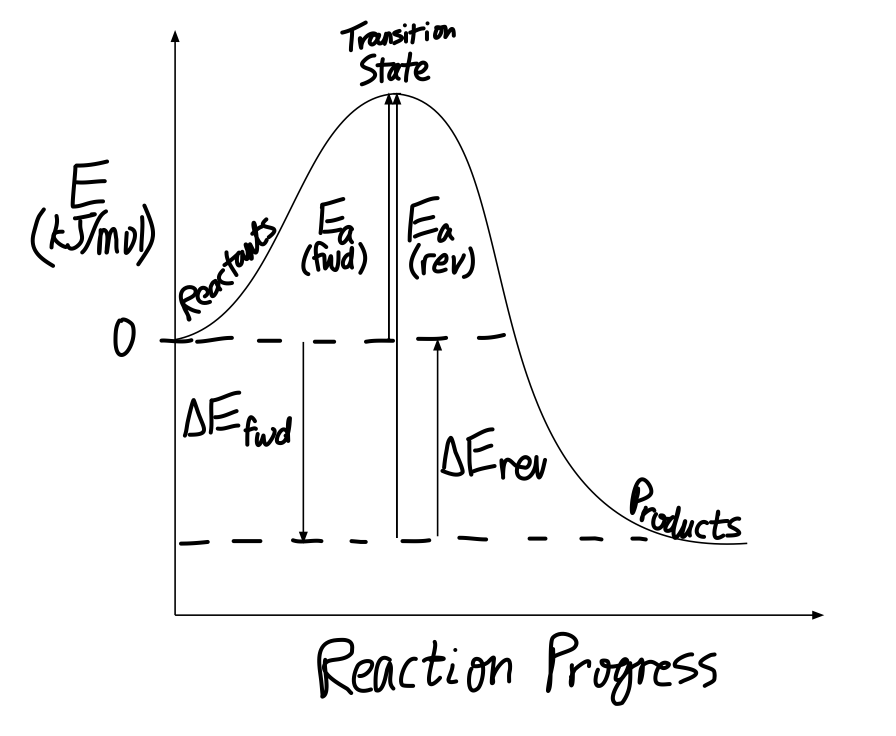
Energy Diagram — Overview & Parts - Expii Energy diagrams typically include: the reactants, transition states, and products. Depending on the reaction, it can also include intermediates and activation energy. Energy diagrams are also referred to as reaction coordinate diagrams! The Different Parts of an Energy Diagram: Image source: Caroline Monahan Labeling an Energy Diagram Diagram | Quizlet Definition Point of reaction in which all bonds between atoms are broken and atoms are free to recombine Location Term Activation Complex Energy Definition Maximum amount of energy required to perform the reaction Location Term Reverse Activation Energy Definition Energy required to break the bonds between atoms for the REVERSE reaction Location 3.7: Energy Diagrams - Physics LibreTexts Elements of Energy Diagrams First of all, it should be noted that we will be confining ourselves to energy diagrams for 1-dimensional motion. This dimension will be represented by the horizontal axis, and the vertical axis will have units of energy. Drag each label to the correct location on the image. Each label can be ... The first diagram (by the left) is an endothermic reaction. In this reaction, the products are higher in energy than the reactants. Heat/Energy is absorbed in this reaction and enthalpy is positive. In second diagram (by the right) is an exothermic reaction. In this reaction, the reactants are higher in energy than the products.
Energy Diagrams - Chemistry LibreTexts In an energy diagram, the vertical axis represents the overall energy of the reactants, while the horizontal axis is the ' reaction coordinate ', tracing from left to right the progress of the reaction from starting compounds to final products. The energy diagram for a typical S N 2 reaction might look like this:
Identify the parts of the energy diagrams - Brainly.com Identify the parts of the energy diagrams 2 See answers Advertisement BarrettArcher Answer : Graph 1 is an endothermic reaction and Graph 2 is an exothermic reaction. The labeling of both the graphs are shown below. Explanation : Endothermic reaction : In this reaction, the energy is absorbed from the surrounding.
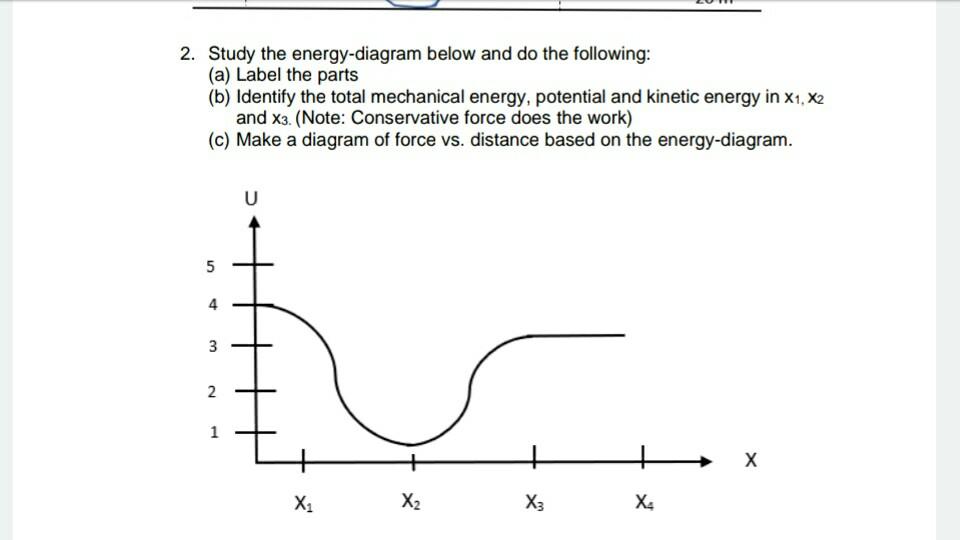
8.5: Potential Energy Diagrams and Stability - Physics LibreTexts The mechanical energy of the object is conserved, E = K + U, and the potential energy, with respect to zero at ground level, is U (y) = mgy, which is a straight line through the origin with slope mg . In the graph shown in Figure 8.5.1, the x-axis is the height above the ground y and the y-axis is the object's energy.
Energy Profile: Definition, Diagram, Reaction | StudySmarter An energy profile shows how the energy of a system changes as the reaction progresses. It is synonymous with an energy diagram/energy profile diagram. As stated previously, the energy profile diagram follows the change in potential energy. The y-axis is the potential energy and the x-axis is the reaction coordinate/reaction progress.
6.7: Energy Diagrams - Chemistry LibreTexts In an energy diagram, the vertical axis represents the overall energy of the reactants, while the horizontal axis is the ' reaction coordinate ', tracing from left to right the progress of the reaction from starting compounds to final products. The energy diagram for a typical one-step reaction might look like this:
AP Chem - 6.2 Energy Diagrams of Reactions | Fiveable In an energy diagram, the vertical axis represents the energy, and the horizontal axis represents the progress of the reaction or phase change. The energy diagram for an endothermic process will have an upward slope, indicating that energy is being absorbed by the system as the reaction or phase change proceeds.
Interpreting a Reaction Energy Diagram | Chemistry | Study.com Steps for Interpreting a Reaction Energy Diagram. Step 1: Label the reactants and the products and determine their energies. Step 2: Identify the activation barrier and its activation energy. Step ...
Energy Diagrams: Definition, Examples, Draw | StudySmarter A potential energy diagram is a graph with enthalpy (potential energy) on the vertical axis and the reaction pathway (time) on the horizontal axis. Consider the following enthalpy, or potential energy, diagrams for a set of hypothetical chemical reactions: i. An exothermic chemical reaction: Figure 4: Enthalpy diagram for an exothermic chemical ...
Potential Energy Diagrams Flashcards | Quizlet is the difference between the potential energy of the reactants and products. The symbol for the heat of reaction is. Delta H. In a chemical reaction, an enzyme acts as a catalyst, causing the. activation energy of the reaction to increase. The only difference between a reaction with a catalyst and a reaction without a catalyst is.
Solved Identify the parts of the energy diagram below: B - Chegg Transcribed image text: Identify the parts of the energy diagram below: B Energy A C Reaction Progress Activation Energy (E₂): [Select] Heat of the Reaction (AHrxn): [Select ] Use the diagram to answer the questions: Is the value of AHrxn positive or negative? [Select ] Is this reaction endothermic or exothermic?
Energy Profiles (Energy Diagrams) Chemistry Tutorial - AUS-e-TUTE From our energy profile diagram we see that 192.4 kJ mol -1 of energy was absorbed by the reactant molecules, but only 100 kJ mol -1 was released as the activated complex broke apart to make the product molecules. Overall, the system absorbed a net amount of energy of 192.4 - 100 = 92.4 kJ mol -1 .

















Post a Comment for "43 identify the parts of the energy diagrams"